
In 1924 the eminent Soviet bio-chemist Alexander Oparin (Алекса́ндр Опа́рин) [1894-1980] published his ground-breaking book entitled ‘The Origin of Life’, within which he examined the theoretical beginnings of life on Earth. In 1922 he stated ‘There is no fundamental difference between a living organism and lifeless matter. The complex combination of manifestations and properties characteristic of life must have arisen as a part of the process of the evolution of matter.’ He asserted that basic organic chemicals might have formed into microscopic localized systems, from which primitive living things could have developed at a very early time in the planet’s existence when conditions were very different to today. Highly volatile and dangerous conditions, according to Alexander Oparin, are exactly the conditions within which amino acids could be produced. Amino acids are protein building blocks of all life. Alexander Oparin went on to develop the field industrial biochemistry for the Soviet State, whilst his earlier work was left unfinished. Alexander Oparin speculated that organic life was a product of natural events and had nothing to do with any form of divine intervention.
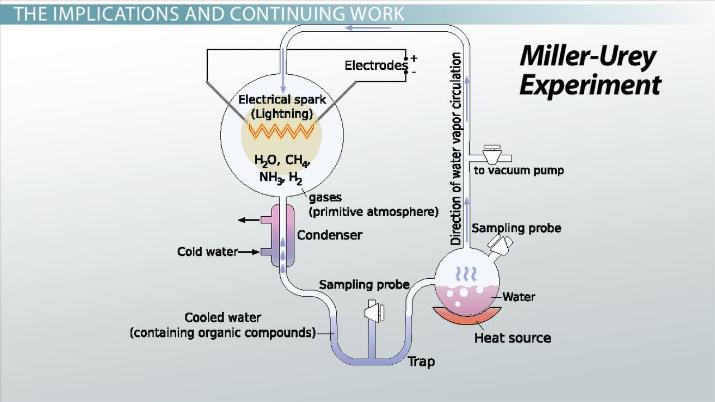
In the United States in 1952, two scientists named Stanley Miller and Harold Urey set about designing an experiment to prove Alexander Oparin correct. This simple but ingenuous experiment involved the use of water (H2O), methane (CH4), ammonia (NH3), and hydrogen (H2). The chemicals were all sealed inside a sterile 5-liter glass flask connected to a 500ml flask which was half-full of liquid water. The liquid water in the smaller flask was heated to induce evaporation, and the water vapour was allowed to enter the larger flask. Continuous electrical sparks were fired between the electrodes to simulate lightning in the water vapour and gaseous mixture, and then the simulated atmosphere was cooled again so that the water condensed and trickled into a U-shaped trap at the bottom of the apparatus.
After one day, the solution collected at the trap had turned pink in colour. At the end of one week of continuous operation, the boiling flask was removed, and mercuric chloride was added to prevent microbial contamination. The reaction was stopped by adding barium hydroxide and sulfuric acid and evaporated to remove impurities. Using paper chromatography, Miller identified five amino acids present in the solution: glycine, α-alanine and β-alanine were positively identified, while aspartic acid and α-aminobutyric acid (AABA) were less certain, due to the spots being faint. At the time results suggested that just a single ‘spark’ of electricity would produce 11 of the commonly known 20 amino acids. Following Miller’s death in 2007, scientists examining the original results of this experiment identified well-over 20 amino acids had been generated entirely due to these replicated ‘primitive’ environmental conditions.
References:
https://www.pbs.org/exploringspace/meteorites/murchison/page5.html
https://ru.wikipedia.org/wiki/Опарин,_Александр_Иванович
